An artificial intelligence approach to create AAV capsids for gene therapies
Adeno-associated viruses (AAVs) have become the go-to vehicle for delivering therapeutic gene cargo to target tissues for the recent wave of gene therapies that are in development in academic and biotechnology laboratories (1). However, natural AAVs do not specifically target diseased cells and tissues, and they can be recognized by the immune system in ways that limit therapeutic success. Synthetic biologists from Dyno Therapeutics, a biotech company pioneering use of artificial intelligence in gene therapy and scientists at Harvard’s Wyss Institute for Biologically Inspired Engineering and Harvard Medical School (HMS) have been taking a “directed evolution” approach to overcome the limitations of current capsids. They have developed new machine-guided technologies to rapidly and systematically engineer a suite of new, improved capsids for widespread therapeutic use. In this machine-guided approach, they randomly mutate specific amino acid building blocks of the capsid proteins that form the shell of the virus and directly contact target cells and evaluate which changes can route the virus to target tissues and successively layering mutations on top of each other in an arduous iterative process.
 Sam Sinai, George Church, Eric Kelsic, and Pierce Ogden are holden small models of the AVVs capsid in their hands. ©Wyss Institute at Harvard University
Sam Sinai, George Church, Eric Kelsic, and Pierce Ogden are holden small models of the AVVs capsid in their hands. ©Wyss Institute at Harvard University
Previous approaches have been limited by the difficulty of altering a complex capsid protein without breaking its function and by the general lack of knowledge regarding how AAV capsids interact with the body. Historically, rather than addressing this challenge directly, the most popular approaches to capsid engineering have taken a roundabout solution: generating libraries of new capsids by making random changes to the protein. However, since most random changes to the capsid actually result in decreased function, such random libraries contain few viable capsids, much less improved ones. Recognizing the limitation of conventionally generated capsid libraries, the authors implemented a machine-guided approach that gathered a vast amount of data using new high-throughput measurement technologies to teach them how to build better libraries and, ultimately, lead to synthetic capsids with optimized delivery properties.
Focusing on the AAV2 capsid, they generated a virus library containing about 200,000 variants and identified capsid changes that both maintained AAV2’s viability and improved its tropism to specific organs in mice. In the process, the authors’ systematic efforts unexpectedly revealed the existence of a previously-unrecognized protein encoded within the sequence of all the most popular AAV capsids, which they termed membrane-associated accessory protein (MAAP). This accessory protein hidden within the capsid-encoding DNA sequence binds to the membrane of target cells. “Membrane-associated accessory protein (MAAP), as we named it, exists in all of the most popular AAV serotypes and we believe that it plays a role in the virus’ natural life cycle, said co-first author Pierce Ogden, a former graduate student and now postdoctoral fellow working with Church. Studying how MAAP functions will be an exciting area for future research and could potentially lead to a better understanding of how to better produce and engineer AAV gene therapies.” These findings are reported in Science (2).
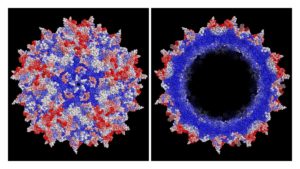 Improved AAV vector capsid for gene therapy engineered with a new machine-guided approach shows, in red, improvements in efficiency of viral production based on the average effect of insertions at all possible amino acid positions, with white showing neutral and blue showing deleterious positions.
Improved AAV vector capsid for gene therapy engineered with a new machine-guided approach shows, in red, improvements in efficiency of viral production based on the average effect of insertions at all possible amino acid positions, with white showing neutral and blue showing deleterious positions.
(Left: capsid viewed from outside, Right: cut-out to reveal inner positions).
©Eric Kelsic, Dyno Therapeutics
“This is just the beginning of machine-guided engineering of AAV capsids to transform gene therapy,” underscores co-author Sam Sinai, Ph.D., Lead Machine Learning Scientist and co-founder of Dyno Therapeutics. “The success of the simple linear models used in this study has led us to pursue more data and higher capacity machine learning models, where the potential for improvement in capsid designs feels boundless.” “The results in the Science publication demonstrate, for the first time, the power of linking a comprehensive set of advanced techniques – large scale DNA synthesis, pooled in vitro and in vivo screens, next-generation sequencing readouts, and iterative machine-guided capsid design – to generate optimized synthetic AAV capsids,” explains co-first and co-corresponding author Eric D. Kelsic, Ph.D., CEO and co-founder of Dyno Therapeutics. The study was funded by Harvard’s Wyss Institute for Biologically Inspired Engineering and the National Institutes of Health.
(1) Our recent report “Gene Therapy Products in Development” (Updated Nov 2019) indicates that AAV-based products make up over 27% of gene therapy products in development in biotech and pharmaceutical companies.
(2) Ogden P.J., Kelsic E.D., Sinai S., Church G.M. Comprehensive AAV capsid fitness landscape reveals a viral gene and enables machine-guided design. Science. 2019 Nov 29;366(6469):1139-43. doi: 10.1126/science.aaw2900
An artificial intelligence approach to create AAV capsids for gene therapies
An artificial intelligence approach to create AAV capsids for gene therapies
Adeno-associated viruses (AAVs) have become the go-to vehicle for delivering therapeutic gene cargo to target tissues for the recent wave of gene therapies that are in development in academic and biotechnology laboratories (1). However, natural AAVs do not specifically target diseased cells and tissues, and they can be recognized by the immune system in ways that limit therapeutic success. Synthetic biologists from Dyno Therapeutics, a biotech company pioneering use of artificial intelligence in gene therapy and scientists at Harvard’s Wyss Institute for Biologically Inspired Engineering and Harvard Medical School (HMS) have been taking a “directed evolution” approach to overcome the limitations of current capsids. They have developed new machine-guided technologies to rapidly and systematically engineer a suite of new, improved capsids for widespread therapeutic use. In this machine-guided approach, they randomly mutate specific amino acid building blocks of the capsid proteins that form the shell of the virus and directly contact target cells and evaluate which changes can route the virus to target tissues and successively layering mutations on top of each other in an arduous iterative process.
 Sam Sinai, George Church, Eric Kelsic, and Pierce Ogden are holden small models of the AVVs capsid in their hands. ©Wyss Institute at Harvard University
Sam Sinai, George Church, Eric Kelsic, and Pierce Ogden are holden small models of the AVVs capsid in their hands. ©Wyss Institute at Harvard University
Previous approaches have been limited by the difficulty of altering a complex capsid protein without breaking its function and by the general lack of knowledge regarding how AAV capsids interact with the body. Historically, rather than addressing this challenge directly, the most popular approaches to capsid engineering have taken a roundabout solution: generating libraries of new capsids by making random changes to the protein. However, since most random changes to the capsid actually result in decreased function, such random libraries contain few viable capsids, much less improved ones. Recognizing the limitation of conventionally generated capsid libraries, the authors implemented a machine-guided approach that gathered a vast amount of data using new high-throughput measurement technologies to teach them how to build better libraries and, ultimately, lead to synthetic capsids with optimized delivery properties.
Focusing on the AAV2 capsid, they generated a virus library containing about 200,000 variants and identified capsid changes that both maintained AAV2’s viability and improved its tropism to specific organs in mice. In the process, the authors’ systematic efforts unexpectedly revealed the existence of a previously-unrecognized protein encoded within the sequence of all the most popular AAV capsids, which they termed membrane-associated accessory protein (MAAP). This accessory protein hidden within the capsid-encoding DNA sequence binds to the membrane of target cells. “Membrane-associated accessory protein (MAAP), as we named it, exists in all of the most popular AAV serotypes and we believe that it plays a role in the virus’ natural life cycle, said co-first author Pierce Ogden, a former graduate student and now postdoctoral fellow working with Church. Studying how MAAP functions will be an exciting area for future research and could potentially lead to a better understanding of how to better produce and engineer AAV gene therapies.” These findings are reported in Science (2).
 Improved AAV vector capsid for gene therapy engineered with a new machine-guided approach shows, in red, improvements in efficiency of viral production based on the average effect of insertions at all possible amino acid positions, with white showing neutral and blue showing deleterious positions.
Improved AAV vector capsid for gene therapy engineered with a new machine-guided approach shows, in red, improvements in efficiency of viral production based on the average effect of insertions at all possible amino acid positions, with white showing neutral and blue showing deleterious positions.
(Left: capsid viewed from outside, Right: cut-out to reveal inner positions).
©Eric Kelsic, Dyno Therapeutics
“This is just the beginning of machine-guided engineering of AAV capsids to transform gene therapy,” underscores co-author Sam Sinai, Ph.D., Lead Machine Learning Scientist and co-founder of Dyno Therapeutics. “The success of the simple linear models used in this study has led us to pursue more data and higher capacity machine learning models, where the potential for improvement in capsid designs feels boundless.” “The results in the Science publication demonstrate, for the first time, the power of linking a comprehensive set of advanced techniques – large scale DNA synthesis, pooled in vitro and in vivo screens, next-generation sequencing readouts, and iterative machine-guided capsid design – to generate optimized synthetic AAV capsids,” explains co-first and co-corresponding author Eric D. Kelsic, Ph.D., CEO and co-founder of Dyno Therapeutics. The study was funded by Harvard’s Wyss Institute for Biologically Inspired Engineering and the National Institutes of Health.
(1) Our recent report “Gene Therapy Products in Development” (Updated Nov 2019) indicates that AAV-based products make up over 27% of gene therapy products in development in biotech and pharmaceutical companies.
(2) Ogden P.J., Kelsic E.D., Sinai S., Church G.M. Comprehensive AAV capsid fitness landscape reveals a viral gene and enables machine-guided design. Science. 2019 Nov 29;366(6469):1139-43. doi: 10.1126/science.aaw2900
 Sam Sinai, George Church, Eric Kelsic, and Pierce Ogden are holden small models of the AVVs capsid in their hands. ©Wyss Institute at Harvard University
Sam Sinai, George Church, Eric Kelsic, and Pierce Ogden are holden small models of the AVVs capsid in their hands. ©Wyss Institute at Harvard University Improved AAV vector capsid for gene therapy engineered with a new machine-guided approach shows, in red, improvements in efficiency of viral production based on the average effect of insertions at all possible amino acid positions, with white showing neutral and blue showing deleterious positions.
Improved AAV vector capsid for gene therapy engineered with a new machine-guided approach shows, in red, improvements in efficiency of viral production based on the average effect of insertions at all possible amino acid positions, with white showing neutral and blue showing deleterious positions.


COMMENTS ARE OFF THIS POST