Arrowhead Pharmaceuticals and Vivo Capital Launch Joint Venture Aimed at Greater China Market Arrowhead Pharmaceuticals, a US company focused on therapies triggering the RNA interference (RNAi), and Vivo Capital, a global healthcare investment firm with offices in Palo Alto, CA, and in China, have formed a new joint venture, Visirna…..
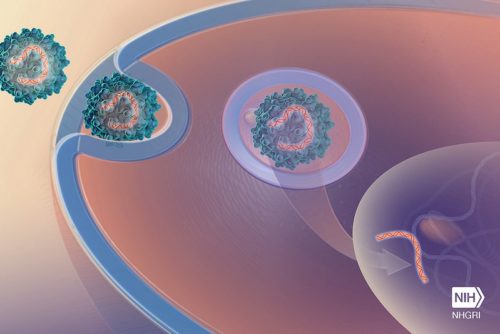
Second phase II study for zilebesiran, an RNAi therapeutic for the treatment of hypertension Alnylam Pharmaceuticals, a leading RNAi therapeutics company, has initiated KARDIA-2 (NCT05103332), a global Phase 2 study to evaluate the efficacy and safety of zilebesiran (formerly known as ALN-AGT), an investigational subcutaneous RNAi therapeutic targeting liver-expressed angiotensinogen…..
Trend Chart on Innovative Bioindustries April 12th FEATURE STORY ● Sirnaomics releases positive interim data from phase II Trial of siRNA candidate studies Sirnaomics, a US-China RNAi therapeutics company, reported on April 9th positive interim results from a Phase II trial of its RNA treatment for non-melanoma skin cancer. STP705…..
More than 580 gene therapy products are under development in biotech and pharmaceutical companies for the treatment of more than 240 diseases. More than 200 of these products are now at the clinical stage. After releasing our « Landscape in… Gene Therapy Companies » in September, we now offer you an opportunity…..
BioPharmAnalyses and OctopusyX BioConsulting are proud to announce the launch of their up-dated and enriched 2019 edition of their report entitled “Landscape… in Gene Therapy Companies”. This report provides you with an in-depth analysis of more than 230 companies involved in this expanding field. (UPDATED SEP 2019) In…..
Gene Therapy is a rapidly and obviously evolving field that is now definitively recognized as a well-acknowledged major medical and scientific area. BioPharmAnalyses is proud to announce the launch of its new global commercial report "Landscape in Gene Therapy Companies". This is the first such report that provides you with an actually updated information and an accurate international mapping…..